WELCOME TO OPEFE ARCHIVES
Piranha Eggs - Serrasalmus spilopleura (= S. maculatus)
with additional information on Larvae and Juveniles: S.
spilopleura v. S. marginatus.
Frank
Magallanes - March 12, 2004
Science
has been studying fish eggs for at least a century, probably longer. China
certainly has the historical knowledge of studying carp eggs as does Medieval
Europe. In those studies up to the present it is well known that higher
temperatures decreases the time for eggs to hatch. Smaller eggs also take less
time to develop than larger eggs (Pauly, D. and Pullin, R. S. V. 1988).
Characin eggs have been studied for a number of years by science. This study
includes the piranhas and pirambebas. Recently, Quagio-Grassiotto and A. C. D.
Guimaraes (2003) discovered the morphology of S. spilopleura (= S.
maculatus) and its egg envelope. The species eggs not only reflects its
oviparous nature, but also its eggs are adhesive. The oocytes layer (acellular
layer) is formed by proteins and polysaccharides. This portion is known as the
zona radiata, zona pellucida, chorinic vitelline envelope, chorion, vitellinic
membrane, or egg envelope. Oviparous species like S. spilopleura, the egg
is structured by primary and secondary envelopes. Much of its creation is by
evolutionary trends, adaptional processes, and environmental conditions (Ivankov
and Kurdyayeva 1973; Nagahama 1983; Yamagami et al. 1992).
In the
subfamily Serrasalminae, which includes the piranhas and pirambebas, genera Pygocentrus,
Serrasalmus, Pristobrycon and Pygopristis, the group have a long
reproductive period during the raining season (Rodrguez et al., 1978; Leao et
al, 1991; Vazzoler and Menezes 1992; Ferreria et al. 1996). Serrasalmus
spilopleura, reproduction is continuous, with multiple spawnings (Lamas and
Godinho 1996). In this study, adult female specimens of S. spilopleura
were caught and collected monthly from the Jurumirim reservoir, Alto
Paranapanema River, Sao Paulo State, Brazil from March 1998 to February 1999.
The specimens were anaesthetized and their gonads were removed, cut into small
segments, and fixed overnight in paraformaldehyde and gluaradehyde in Sorensen
phosphate buffer. The ovulated eggs were removed from the ovarian cavity and
fixed the same way. Sections of prepared ovary and ovulated eggs were examined
and photographed using an electron microscope.
Below,
is the abstract on this study:
Abstract
Quagio-Grassiotto,
I. and Guimarăes, A. C. D. 2003. Follicular epithelium, theca and egg envelope
formation in Serrasalmus spilopleura (Teleostei, Characiformes,
Characidae). Acta Zoologica (Stockholm) 84: 121.
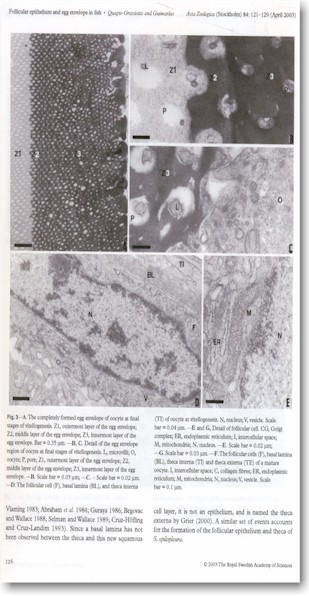 The follicular epithelium and theca of oocytes in Serrasalmus
spilopleura differentiates during the initial primary growth phase. The
follicular cells are squamous and the thecal cells are disposed in two layers.
During the secondary growth phase, follicular cells become cuboidal, acquire
characteristics typical of protein- or glycoprotein-producing cells, and show
dilated intercellular spaces. Formation of the egg envelope in S. spilopleura
begins in the previtellogenic oocytes as a layer of amorphous electron-dense
material is laid down on the oolemma. During vitellogenesis, another layer of
electron-dense material appears beneath the first layer. Also during this phase,
a layer of amorphous, less electron-dense material is formed adjacent to the
follicular epithelium. The secondary egg envelope appears at the
postvitellogenic phase and is composed of a filamentous and undulant material.
The morphology of the egg envelopes in S. spilopleura reflects not only
its oviparous nature but also the fact that its eggs are adhesive.
The follicular epithelium and theca of oocytes in Serrasalmus
spilopleura differentiates during the initial primary growth phase. The
follicular cells are squamous and the thecal cells are disposed in two layers.
During the secondary growth phase, follicular cells become cuboidal, acquire
characteristics typical of protein- or glycoprotein-producing cells, and show
dilated intercellular spaces. Formation of the egg envelope in S. spilopleura
begins in the previtellogenic oocytes as a layer of amorphous electron-dense
material is laid down on the oolemma. During vitellogenesis, another layer of
electron-dense material appears beneath the first layer. Also during this phase,
a layer of amorphous, less electron-dense material is formed adjacent to the
follicular epithelium. The secondary egg envelope appears at the
postvitellogenic phase and is composed of a filamentous and undulant material.
The morphology of the egg envelopes in S. spilopleura reflects not only
its oviparous nature but also the fact that its eggs are adhesive.
S.
spilopleura, the thickness of the eggs and the primary envelope is involved
in the process of fertilization. This protects the integrity of the egg until
the embryo has completely developed (Yamagami et al. 1992). This thickness of
the egg is related to different reproductive strategies. Therefore, the egg
thickness is subject to heavy mechanical stress than in buoyant eggs or those
laid in sheltered areas (Ivankov and Kurdyayeva 1973). S. spilopleura
shows an inherent thickness in its primary envelope which is likely related to
is oviparous nature.
 Like other Serrasalminae, their eggs are
laid on submerged marginal vegetation (Cutright 1942). In the work, Adhesiveness
and surface patterns of eggs in neotropical freshwater teleosts (Rizzo, Sato,
Barreto, and Godinho, 2002) the eggs of S. spilopleura were found to be
honeycomb-like pores. This special arrangement of the outer zona radiata was
wider than those other species examined in this study. S. spilopleura eggs
were found to be polygonal in shape and organized in a honeycomb fashion. Of
special interest the mycropylar disc of S. spilopleura resembled the
adhesive disc of Pygocentrus nattereri (Wirz-Hlavacek & Riehl, 1990).
In S. spilopleura this apparatus does not contain filaments.
Like other Serrasalminae, their eggs are
laid on submerged marginal vegetation (Cutright 1942). In the work, Adhesiveness
and surface patterns of eggs in neotropical freshwater teleosts (Rizzo, Sato,
Barreto, and Godinho, 2002) the eggs of S. spilopleura were found to be
honeycomb-like pores. This special arrangement of the outer zona radiata was
wider than those other species examined in this study. S. spilopleura eggs
were found to be polygonal in shape and organized in a honeycomb fashion. Of
special interest the mycropylar disc of S. spilopleura resembled the
adhesive disc of Pygocentrus nattereri (Wirz-Hlavacek & Riehl, 1990).
In S. spilopleura this apparatus does not contain filaments.
Despite
the the absence of data that elucidate the mechanism by which eggs of these
species adhere to substrata, it could be postulated that the micropylar disc may
play a role in adhesiveness. Since the micropyle in these fishes is located in
the center of the micropylar disc, synchronization of male and female spawning
behavior is of striking importance (Riehl & Appelbaum, 1991). In this case,
the eggs should be fertilized before they attach to the substratum as in P.
nattereri. In conclusion, the egg surface pattern is related to the degree
of egg adhesiveness and is the same at the gender level in Characiformes; on the
other hand, there is a strong correlation between jelly coat and Siluriformes
eggs, apparently without a relationship between structure and adhesiveness.
 Another aspect is the morphometric
variation of larvae and juveniles of Serrasalmus spilopleura (= S.
maculatus) and S. marginatus of the Paraná basin, Brazil. Maristela
Cavicchioli, Keshiyu Nakatani and Oscar Akio Shibatta (1997). The larvae and
juveniles of Serrasalmus spilopleura and S. marginatus of the
upper Paraná river floodplains are described. The character that distinguishes
the species in the early stages of larval development in the number of myomeres:
32-35 (mean 34.56) for S. spilopleura and 36-39 (37.01) for S.
marginatus. The shape of the snout, the postmedian region, and the caudal
peduncle were determinant in the separation of the species in the flexion and
principal components analysis, allied to the geometrical method of shape
analysis, discriminated morphometrically the species at all developmental
stages. The length of the snout and the distances related to the postmedian
region and caudal peduncle distinguished the species in all the series of
landmarks and were, therefore, fundamental discriminating variables for the
larval stages and juvenile period.
Another aspect is the morphometric
variation of larvae and juveniles of Serrasalmus spilopleura (= S.
maculatus) and S. marginatus of the Paraná basin, Brazil. Maristela
Cavicchioli, Keshiyu Nakatani and Oscar Akio Shibatta (1997). The larvae and
juveniles of Serrasalmus spilopleura and S. marginatus of the
upper Paraná river floodplains are described. The character that distinguishes
the species in the early stages of larval development in the number of myomeres:
32-35 (mean 34.56) for S. spilopleura and 36-39 (37.01) for S.
marginatus. The shape of the snout, the postmedian region, and the caudal
peduncle were determinant in the separation of the species in the flexion and
principal components analysis, allied to the geometrical method of shape
analysis, discriminated morphometrically the species at all developmental
stages. The length of the snout and the distances related to the postmedian
region and caudal peduncle distinguished the species in all the series of
landmarks and were, therefore, fundamental discriminating variables for the
larval stages and juvenile period.
REFERENCES
-
Cavicchioli,
M., Nakatani, K., and Shibatta, O. A. 1997, Ichthyol. Explor Freshwaters, Vol.
8, No. 2, pp. 97-106.
-
Quagio-Grossioto,
I., and Guimarăes, A. C. D. 2003, Acta Zoologica (Stockholm) 84: 121-129
(April 2003).
-
Yamagami,
K., Hamazaki, T. S., Yasumasu, S. and Masuda, K., Luchi, I. 1992. Molecular
and cellular basis of formation, hardening, and breakdown of the egg envelope
in fish. - International Review of Cytology 136: 51.92.
-
Ivankov,
V. N. and Kurdyayeva, V. P., 1973. Systemic differences and ecological
importance of the membranes in fish eggs. - Journal of Ichthyology 13:
864-873.
-
Pauly,
D. and Pullin, R. S. V. 1988. Hatching time in spherical, pelagic, marine fish
eggs in response to temperature and egg size. Environ. Biol. Fish. 22(4):
261-271.
-
Breder,
C. M. Jr. and Rosen, D. E. 1966. Modes of reproduction in fishes. T.F.H.
Publications, Neptune City, NJ. 941 p.
-
Dannevig,
H. 1895. The influence of temperature on the development of the eggs of
fishes. Rep. Fish. Board Scotland 1894: 147-152.
-
Rizzo.,
Sato, Y., Barreto B. P., and Godinho, H. P. 2002, Journal of Fish Biology, 61,
615-632.
-
Wirz-Hlavacek,
G. & Riehl, R. 1990. Reproductive behavior and egg structure of the
piranha Serrasalmus nattereri (Kner, 1860), Acta Biologia. Berodis 2,
19-38.
USE YOUR BACKSPACE TO RETURN OR CLICK HERE TO
RETURN RESEARCH PAGE
TO
RETURN HOME CLICK HERE
The OPEFE web site and its contents; is disclaimed for purposes of
Zoological Nomenclature in accordance with the International Code of Zoological
Nomenclature, Fourth Edition, Article 8.3 and 8.4. No new names or nomenclature
changes are available from statements at this web site.
Copyright© 1994-2012 Oregon Piranha Exotic Fish Exhibit (The OPEFE
fish exhibit is permanently CLOSED as of 2000) Sutherlin, Oregon. Information
posted on this web site is archival data on fish scientific classifications and
other information. DISCLAIMER: The copyrighted material may not be used
for any purpose other than private study, scholarship or research. Cited
information requires credit and this link www.opefe.com.
All rights reserved. All images shown (unless otherwise noted) is property
of OPEFE.

 The follicular epithelium and theca of oocytes in Serrasalmus
spilopleura differentiates during the initial primary growth phase. The
follicular cells are squamous and the thecal cells are disposed in two layers.
During the secondary growth phase, follicular cells become cuboidal, acquire
characteristics typical of protein- or glycoprotein-producing cells, and show
dilated intercellular spaces. Formation of the egg envelope in S. spilopleura
begins in the previtellogenic oocytes as a layer of amorphous electron-dense
material is laid down on the oolemma. During vitellogenesis, another layer of
electron-dense material appears beneath the first layer. Also during this phase,
a layer of amorphous, less electron-dense material is formed adjacent to the
follicular epithelium. The secondary egg envelope appears at the
postvitellogenic phase and is composed of a filamentous and undulant material.
The morphology of the egg envelopes in S. spilopleura reflects not only
its oviparous nature but also the fact that its eggs are adhesive.
The follicular epithelium and theca of oocytes in Serrasalmus
spilopleura differentiates during the initial primary growth phase. The
follicular cells are squamous and the thecal cells are disposed in two layers.
During the secondary growth phase, follicular cells become cuboidal, acquire
characteristics typical of protein- or glycoprotein-producing cells, and show
dilated intercellular spaces. Formation of the egg envelope in S. spilopleura
begins in the previtellogenic oocytes as a layer of amorphous electron-dense
material is laid down on the oolemma. During vitellogenesis, another layer of
electron-dense material appears beneath the first layer. Also during this phase,
a layer of amorphous, less electron-dense material is formed adjacent to the
follicular epithelium. The secondary egg envelope appears at the
postvitellogenic phase and is composed of a filamentous and undulant material.
The morphology of the egg envelopes in S. spilopleura reflects not only
its oviparous nature but also the fact that its eggs are adhesive.